Editorial Car T Cells - What Have We Learnt? Nature Reviews Clinical Oncology 15 1 (2017)
- Review
- Open Admission
- Published:
Chimeric antigen receptor T prison cell (Machine-T) immunotherapy for solid tumors: lessons learned and strategies for moving forwards
Journal of Hematology & Oncology volume eleven, Article number:22 (2018) Cite this article
Abstract
Recently, the US Food and Drug Administration (FDA) approved the kickoff chimeric antigen receptor T jail cell (CAR-T) therapy for the treatment CD19-positive B cell acute lymphoblastic leukemia. While CAR-T has accomplished remarkable success in the treatment of hematopoietic malignancies, whether it can do good solid tumor patients to the same extent is even so uncertain. Even though hundreds of clinical trials are undergoing exploring a variety of tumor-associated antigens (TAA), no such antigen with comparable properties similar CD19 has yet been identified regarding solid tumors CAR-T immunotherapy. Inefficient T jail cell trafficking, immunosuppressive tumor microenvironment, suboptimal antigen recognition specificity, and lack of prophylactic control are currently considered as the primary obstacles in solid tumor CAR-T therapy. Here, we reviewed the solid tumor CAR-T clinical trials, emphasizing the studies with published results. Nosotros farther discussed the challenges that CAR-T is facing for solid tumor handling and proposed potential strategies to improve the efficacy of Auto-T equally promising immunotherapy.
Background
Recently, the US Food and Drug Assistants (FDA) approved the start chimeric antigen receptor T jail cell (Motorcar-T) therapy for the handling of children and young adults with relapsed or refractory B cell acute lymphoblastic leukemia (ALL) positive for CD19 antigen [one, 2]. Chimeric antigen receptors (CARs) are chimeric immunoglobulin T jail cell receptor (TCR) molecules derived from transgenes encoding for single-concatenation variable fragments (scFv), which originate from antibodies capable of recognizing tumor-associated antigens (TAA) [3, 4]. Mechanistically, the Automobile-T cell recognizes and binds to TAA, inducing a conformational modify that transmits the bounden signal into the Motorcar-T cell. Activation point through the CD3ζ domain and costimulatory domains activate Automobile-T cell, leading to cytokine release and transcription cistron expression, which promote T cell survival and function and eventually induce cytotoxic activities against tumor cells [5, 6]. The molecular mechanisms of Motorcar-T immunotherapy were summarized in Fig. 1.
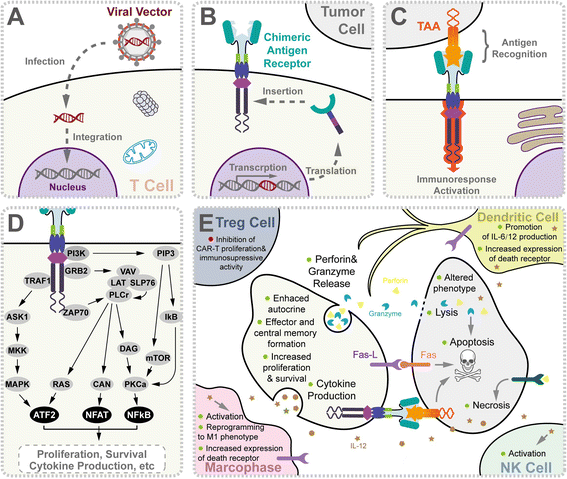
Molecular mechanism of chimeric antigen receptor T prison cell-mediated antitumor activity. a The chimeric T cell receptor coding sequence is delivered by viral vector. Afterwards entering into T cells (beige), virus was uncoated and transgene was preferably integrated at genome transcriptional kickoff sites using specific vector designs, such as MLV retrovirus and piggyBac transposon. b CAR transgenes were endogenously transcript by host mechanism, translated, and later inserted onto the T prison cell surface. c Association of CARs to TAA (orange) on tumor prison cell surface activates T cell for immunological response, for instance, signaling network of CAR-T composed of CD8-CD28-CD137-CD3ζ domains was shown in (d). Motorcar-T-mediated immune response was reported to exist amplified by ZAP70, TRAF1, PI3K, and GRB2 equally well as other uncharacterized factors, giving ascension to upregulation of signaling intermediates and subsequent pro-decease gene transcriptions. e Upon CAR activation, T cells secreted cytokines (brown), perforins (bright yellow), and granzymes (blue) also equally activated decease receptors, which triggered downstream targets. These subcellular events directly or subsequently contribute to specific death of tumor cells, including perforin and granzyme release, cytokine production, direct lysis, apoptosis, necrosis, reprogrammed phenotype, and immuno-memory formation in T cells, tumor cells (gray), macrophages (pinkish) (via IL-half dozen, IL-ten, IL-12, MCP-ane, IP-10, TNF-α, MIP-1α, MIP-1β, IFN-γ), NK cells (cyan) (via IL-12, TNF-α, IFN-γ), Treg cells (navy) (via IL-two, IL-four, IL-7, IL-12, IL-xv, IFN-β, IFN-γ, TSLP), and dendritic cells (yellow) (via IL-half-dozen, IL-ten, IL-12, TNF-α, MIP-1α, MIP-1β, IFN-γ). Abbreviation: NK cells natural killer cells, TAA tumor-associated antigen, Treg cells regulatory T cells
The designs of CARs are grouped schematically into three generations with increasing costimulatory activity [7]. The kickoff generation CARs are conjugated with TCR-CD3ζ concatenation alone, which is capable of providing a comparable stimulatory signal to that of the unabridged CD3 complex [viii, nine]. Still, this Car configuration is insufficient to prime number resting T cells for proliferation and cytokine production, affecting sustained antitumor responses in vivo [10]. With the aim to raise the stimulation effect, the second-generation CARs include a costimulatory module on the basis of the offset generation, which was initially designed in the 1990s [viii, nine, 11, 12]. CD28 is one of the about commonly utilized molecules for this purpose to promote interleukin-2 (IL-2) secretion and meliorate T cell activeness [13,xiv,xv,16]. On top of CD3ζ and co-stimulators like CD28, additional costimulatory domains, such as OX40 or 4-1BB, were added to the tertiary generation CARs to farther enhance the signaling capacity [17, 18]. The fourth-generation CARs added IL-12 to the base of the 2nd-generation constructs, which are known as T cell redirected for universal cytokine-mediated killing (TRUCKs). TRUCKs augment T cell activation and activate and attract innate immune cells to eliminate antigen-negative cancer cells in the targeted lesion. Such TRUCK T cells can also treat viral infections, metabolic disorders, and auto-allowed diseases [19,20,21].
Whereas ongoing CAR-T clinical trials for the treatment of leukemia and lymphoma have demonstrated durable remission of the illness or even cure, Motorcar-T therapy targeting solid tumor is yet in an babe stage. Ane of the virtually frequently asked questions is whether Machine-T can benefit solid tumor patients to the same extent every bit it does for blood malignancies. Hither, we reviewed the published results of clinical studies for solid tumor Machine-T treatment. Nosotros further discussed the challenges that Machine-T is facing for solid tumor treatment and proposed potential strategies to ameliorate the efficacy of Auto-T equally promising immunotherapy.
Clinical trials using engineered CAR-T cells to treat solid tumor
Because of the success achieved in CAR-T therapy targeting B prison cell malignancies and the advancements in CAR-T preclinical studies for solid tumors, more 100 Car-T clinical trials targeting solid tumors accept been initiated at medical centers all over the globe (Table 1).
Tumor-associated antigens and Machine design
So far, no such cell surface antigen with comparable properties as CD19 has all the same been identified regarding solid tumors. An platonic molecule for Motorcar targeting should be overexpressed on cancer prison cell surface of many patients, with zero or very low expression in normal tissues. Currently, TAAs, including mesothelin (MSLN), HER2, EGFR/EGFRvIII, GD2, CEA, IL13Rα2, MUC1, FAP, PSMA, and PSCA, are extensively investigated in CAR-T therapy for solid tumors [22, 23]. TAAs currently existence exploited for Automobile-T therapy in solid tumors are summarized (Fig. 2). Yu and colleagues comprehensively discussed these antigens regarding their biological functions and antitumor activities [22]. Equally shown in Table 1, most of the solid tumor Automobile-T clinical trials use the second or third generation of CARs, which contain either CD28 lone or CD28-4-BB1/OX40 as the costimulatory signal. Notably, a few of these studies, due east.g., the trials targeting GD2 (NCT02765243, NCT02992210), PMSA (NCT03185468), FR-α (NCT03185468), investigated the efficacy of the fourth-generation CARs, i.east., TRUCK, which includes a transgenic cytokine expression cassette in the CAR constructs [24]. Because of the tremendous phenotypic multifariousness in solid tumor lesions, a reasonable number of cancer cells are not recognized past a given Auto. The introduction of a transgenic cytokine such as interleukin-12 (IL-12) initiates universal cytokine-mediated killing towards those cancer cells that are invisible to CAR-T cells [19].
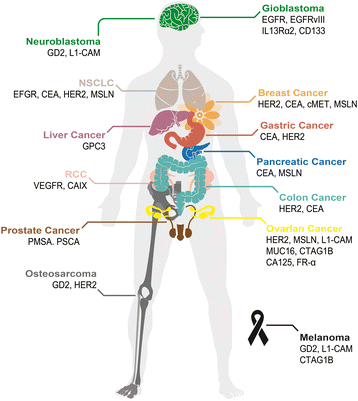
Tumor-associated antigens targeted in CAR-T therapy. Schematic illustration of a human being body whose tissues or organs accept been investigated in preclinical and clinical studies for solid tumor immunotherapy using CAR-T, including brain (green), lungs (biscuit), mammary gland (orangish), liver (purple), stomach (carmine), pancreas (blue), kidneys (pink), colon (cyan), male reproductive system (chocolate-brown), ovary (yellow), and bones (gray) likewise as skin (blackness). Abbreviation: CCA cholangiocarcinoma, MPM malignant pleural mesothelioma, NSCLC non-small-cell lung carcinoma, RCC renal cell carcinoma
CAR-T cell therapy must carefully residuum efficient T cell activation, to ensure antitumor activity, with the potential for uncontrolled cytotoxicity. Some recent clinical trials included an inducible caspase 9 (iCasp9) "safety switch" in their CAR construct, which allows for the removal of inappropriately activated CAR-T cells [25, 26]. The administration of the small-scale molecule drug AP1903 causes the dimerization and activation of iCasp9, resulting in rapid consecration of apoptosis in transduced cells. 2 CAR-T clinical trials targeting GD2 performed at Baylor Higher of Medicine incorporated such iCasp9 switch into their tertiary generation CAR constructs (NCT01822652, NCT02439788). In another recently initiated trial led by Chang, a 4th-generation CAR was fused with the iCasp9 domain (NCT02992210).
PD-L1 expression in solid tumors counteracts the efficacy of CAR-T cell [27]. To overcome the inhibitory outcome of PD-LI expression on Car-T therapy, some clinical trials (eastward.yard., NCT03030001, NCT03182803-MSLN, NCT03182816, NCT02873390, NCT02862028-EGFR, NCT03170141-EGFRvIII, and NCT03179007-MUC1) add PD-1 ascendant-negative receptor expressing gene to CAR-T cells, providing cell-intrinsic checkpoint occludent and increase antitumor efficacy. Based on the currently available data, anti-PD-ane combination therapy approach may exist useful to augment CAR-T cell efficacy and persistence in patients [27].
T prison cell dosage, administration, and persistence
According to the published results and clinical trial description available on the website, the majority of the Motorcar-T clinical trials employ a dose escalation regime, which usually covers two log steps with a starting point ranged from around 1 × 10half dozen and ane × xnine CAR-T cells [24]. It is important to keep in heed that the percent of Machine-positive T cells varies significantly not only among different trials just too among different batches within unmarried trial [24]. I of the major sources for such variation is the dissimilar production procedure of Automobile-T cells adopted past different trials, emphasizing the importance of standardizing the Motorcar-T cells production protocol. Notably, to increase the tolerability of the handling and to lower the risk of side effects, the given Auto-T cell dose is oftentimes split over multiple injections. For example, Ahmed and colleagues reported administration of 1 × x4–1 × 108/m2 HER2 specific CAR-T cells in up to 9 infusions (NCT00902044) [28]. Typically, Automobile-T cells are infused intravenously. Yet, intratumoral (NCT02587689) [29], intracranial (NCT00730613) [xxx], hepatic avenue (NCT01373047) [31], and pleural [32] administration are being investigated besides. The Automobile-T cell administration method could have a dramatic affect on the efficacy of the treatment. Brown et al. compared the effectiveness of ii intracranial CAR-T cell commitment route-infusion into the resected tumor cavity and infusion into the ventricular system [33]. While intracavitary therapy appeared to control local tumor recurrence, progression of glioblastoma at distant sites was observed. Past contrast, intraventricular administration of Machine-T cells leads to the regression of all central nervous system tumors, including spinal tumors. Later on the administration, CAR-T cell persistence is a key gene that determines the efficacy of the therapy. One major trouble of current CAR-T jail cell immunotherapy is that T lymphocytes have limited replicative lifespans [34], which potentially influences the long-term antitumor upshot of Car-T cell immunotherapy. The reported T cell persistence in published clinical trials ranged from upwards to 4 weeks (DDHK97-29/P00.0040C, BB-IND 12084) to up to 192 weeks (NCT00085930).
Clinical outcomes and toxicities
Some early attempts using Motorcar-T to treat solid tumors were not successful. In 2006, Kershaw and colleagues tested the efficacy of CAR-T cells targeting ovarian cancer cells expressing alpha-folate receptor (FR-α) (NCT00019136) [35]. Fifty-fifty though the patients tin can tolerate the assistants of FR-α specific Motorcar-T cells, no clinical response was observed in all the patients (14 out of 14), probable considering of the inefficient localization and short-term persistence of the CAR-T cells [35]. Lamers and colleagues treated 12 patients with CAIX-expressing metastatic renal jail cell carcinoma with CAIX specific CAR-T cells [36, 37]. Unfortunately, no clinical responses were observed, and some patients adult anti-CAR-T jail cell antibodies and severe live enzyme disturbance.
Park et al. reported treating metastatic neuroblastoma with L1-CAM specific Machine-T cells at a dosage of 1 × 10eight cells/chiliadii to i × xix cells/mii (NCT00006480) [38]. The persistence of cells was measured to be 1–seven days for patients with heavy affliction burden and 42 days for a patient with lite disease brunt. One of the six enrolled patients achieved stable disease after treatment [38]. A series of CAR-T clinical trials targeting MSLN take been performed in many different types of solid tumors, such as mesothelioma (NCT01355965), pancreatic cancer (NCT01583686, NCT02465983, NCT02706782), and breast cancer (NCT02792114). In one of these trails, advanced mesothelioma patients were administered autologous T cells electroporated with the mRNA encoding for MSLN Car (NCT01355965) [39, 40]. Moderate clinical responses were observed equally supported by the detection of MSLN specific Motorcar-T cells in the tumor site and the transient superlative of inflammatory cytokines [39, 40]. MSLN CAR T prison cell infusions were well tolerated at the dosage tested without severe toxicities [39, 40]. Unfortunately, 1 of the study subjects was reported to develop severe anaphylaxis and cardiac arrest subsequently the third infusion of MSLN Auto-T cells [40], which is probable to result from the inclusion of the murine SS1 scFv in the Machine-T blueprint. In another trial for pancreatic ductal adenocarcinoma (PDAC) (NCT01897415), Beatty and colleagues utilized mRNA-encoded mesothelin-specific CAR to treat vi patients, in which the infusion were well tolerated and preliminary evidence of antitumor efficacy was observed, supported by stable disease seen in 2 treated patients [41].
Clinically, the safety and efficacy of HER2-specific Auto-T cells in patients with relapsed/refractory HER2-positive sarcoma has been evaluated in a stage I/II clinical report (NCT00902044, NCT01109095, and NCT00924287). In a dose escalating trial (NCT00902044, ane × 104/k2 to 1 × 10viii/mtwo), iv out of nineteen subjects acquired stable disease [28]. Feng et al. performed a phase I clinical trial investigating HER2-specific CAR-T cells in patients with advanced biliary tract cancers (BTCs) and pancreatic cancers (PCs) (NCT01935843). Amongst the 6 patients received HER2-specific Motorcar-T infusion, 1 patient obtained a four.five-month fractional response and the other 5 patients accomplished stable disease with mild to moderate adverse events [42]. In some other trial (NCT01109095), 17 CMV-seropositive patients with radiologically progressive HER2-positive glioblastoma were infused with HER2 CMV bispecific Automobile-T cells at the dose of ane × 106/mtwo–1 × 108/chiliadii [43]. Such treatments were well tolerated without astringent adverse events or cytokine release syndrome, and 7 out the 17 treated patients accomplished stable disease [43].
Feng and colleagues reported the clinical trials of EGFR specific Automobile-T treating non-small prison cell lung cancer (NCT01869166) [44]. Five out of the 11 treated patients achieved stable disease and two achieved partial response. The CAR-T infusion-related adverse events were mild and manageable. Recently, Feng et al. reported treating a patient diagnosed equally advanced unresectable/metastatic cholangiocarcinoma (CCA) with Machine-T cocktail immunotherapy, which was composed of successive infusions of CAR-T cells targeting epidermal growth factor receptor (EGFR) and CD133 (NCT01869166, NCT02541370) [45]. The patient achieved an 8.v-month partial response (PR) from the CART-EGFR therapy and a iv.five-month-lasting PR from the CART133 handling. However, a serial of agin events were besides observed in the course of treatment, including deteriorative grade 3 systemic subcutaneous hemorrhages and congestive rashes together with serum cytokine release.
In 2015, Dark-brown et al. reported that three patients with recurrent glioblastoma were treated with CAR-T cells targeting IL13Rα2 (NCT00730613) [thirty]. Patients received up to 12 local infusions at a maximum dose of 1 × 108 CAR-T cells. Evidence for transient antitumor activity was observed in two of the patients with manageable temporary primal nervus system inflammation. The same group conducted another clinical trial (NCT02208362), in which tumor regression and increased production of cytokines and immune cells were observed [33]. Encouragingly, the clinical response continued for 7.5 months after the initiation of CAR-T cell therapy, indicating a relatively long persistence of CAR-T cells.
Katz and colleagues conducted a phase I trial to test CAR-T in patients with CEA-positive liver metastases (NCT01373047). Among the half-dozen patients who completed the protocol, 1 patient remained alive with stable illness at 23 months post-obit Automobile-T treatment and five patients died of progressive disease [31]. Biopsies demonstrated an increase in liver metastases necrosis or fibrosis in four out of vi patients. No patient suffered a grade 3 or 4 adverse result related to the CAR-T treatment. Louis and colleagues evaluated the efficacy of GD2-specific Automobile-T in nineteen patients either with remission (viii patients) or progressive neuroblastoma (11 patients) (NCT00085930) [46]. 3 of eleven patients with active disease achieved complete remission and up to 192 weeks of CAR-T cell persistence was observed in the trial [46].
Clinical trials investigating the efficacy of CAR-T cells targeting MUC1 (NCT02587689) [29], cMet (NCT01837602, NCT03060356) [47], PSMA (BB-IND 12084) [48], VEGFR-two (NCT01218867) have been reported. Relevant data and results of CAR-T clinical trials were summarized in Table 2.
Equally learned from the results of these clinical trials, Motorcar-T cells face a unique set up of challenges in the case of solid tumors. Some of the issues announced to be the absence of unique tumor-associated antigens, the inefficient homing of T cells to tumor sites, and the limited persistence of CAR-T cells. Moreover, the immunosuppressive microenvironment within the tumor tends to inhibit CAR-T cell function strongly. While a seemingly complicated, fulfilling all of above requirements tin can be accomplished efficiently through both intrinsic and/or extrinsic modifications of Machine-T cells.
Overcoming challenges with smarter Motorcar designs
Different from B cell malignancies, the application of the CAR-T prison cell strategy to non-hematopoietic cancer is faced with physical barriers as well as a variety of approaches that tumors employ to blunt host immune-surveillance [49]. Therefore, obstruction factors existing in current Auto-T trials such as fibrosis, inflammation, autoimmunity, T jail cell exhaustion and persistence, or recurrence must exist overcome with smarter redirected T cell designs to achieve optimal therapeutic results (Fig. iii).
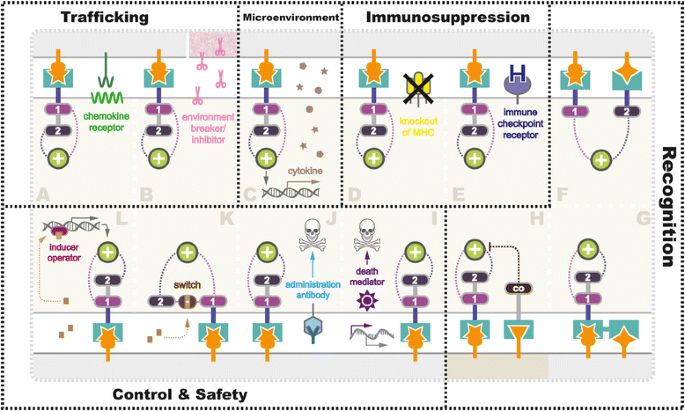
Strategies being exploited to overcome challenges in CAR-T therapy in solid tumor. Diverse strategies are currently existence tested in preclinical and clinical studies to overcome the challenges facing Machine-T therapy for solid tumor (grey), including a, b enhancing T cell (biscuit) trafficking, c reforming tumor microenvironment (pink represented physiological barriers), d, east anti-immunosuppression, elevating antigen recognition towards tumor (f, 1000), and healthy cells (darker beige) (h), also as i–l improving rubber control using suicide switch or on-switch. Detailed mechanisms were further illustrated in the section "Overcoming Challenges with Smarter CAR Designs"
Enhancing trafficking
Tumors are organized tissues with numerous connections with surrounding cells [50], including resident allowed system, i.east., myeloid compartment and lymphoid compartment [51], vasculature, fibroblasts, signaling molecules, and the extracellular matrix [52]. In addition, the low pH, low oxygen, low nutrients condition, and the tumor surrounding tissues, especially blood vessels and fibroblasts, make therapeutic delivery cruelly difficult [53].
In current trials, homing of Car-T cells to the tumor sites is often inefficient attributable to the reasons mentioned above. The successful application of CAR-T cells confronting hematological malignancies is at least partially attributed to the fact that tumor and effector T cells tended to migrate to similar sites due to their shared hematopoietic origins. Solid tumors are known to secrete immuno-factors, including chemokine, cytokine, and growth factors, preventing effector T cells from infiltrating into the tumor bed (Fig. 3a). In 2010, a recombinant chemokine receptor ligand CCR2(b) was introduced into CAR-T cells targeting MSLN [54], and GD2 [55], because the endogenous chemokine receptors on T cells fairly mismatch with the chemokines secreted by the tumors. Beyond minimizing the effects of chemokine, strategies targeting activated surrounding fibroblast (fibroblast activation protein, FAP) in mouse models of mesothelioma and lung cancer [56, 57], vasculature (anti-VEGFR-2) in melanoma, and renal cancer (NCT01218867) [58], as well as tumor stroma (heparanase) in neuroblastoma mice [59], have been tested.
Bereft T cell trafficking is a major functional claiming in anti-cancer immunotherapy. Little has been improved regarding T cell infiltration in the past decade, which is primarily due to our limited understanding of the tumor surrounding compartments and their effects on Machine-T cells [51]. More tumor-associated biomarkers related to T cell filtration should be investigated, such as alpha smooth muscle actin, Thy-1, desmin, and S100A4 protein [lx]. In addition, strategies adult in non-Motorcar-T therapy context might also exist benign to CAR-T blueprint and utilized for penetrating the concrete bulwark and thus enhancing CAR-T cell trafficking. Beyer et al. described a self-dimerizing epithelial intercellular junction opener JO-1 that bound to desmoglein two [61]. Manipulation of JO-one transiently broke tight junctions in polarized epithelial tumor cells, resulting in increased efficiency in mAb treatments (trastuzumab and cetuximab) in xenograft tumor models. Moreover, intratumoral relaxin expression was reported to degrade extracellular matrix poly peptide around solid tumors transiently, which enhanced trastuzumab treatment [62] (Fig. 3b). Non-signaling extracellular hinge domain of CAR-T has too been shown to exhibit an affect on the migratory chapters of CAR-T cells. Qin et al. generated 2 versions of Auto vectors, with or without a swivel domain, targeting hematopoietic, and solid tumor antigens [63]. The CARs with a hinge domain demonstrated better expansion and migration capacity in vitro and Car-T cells expressing anti-mesothelin CARs containing a hinge domain showed enhanced antitumor activities.
Reforming microenvironment
Malignant transformation and the growth of tumor mass influence surrounding microenvironment, which subsequently gives rise to peripheral immune tolerance [64]. In return, tumor microenvironment provides a driving forcefulness contributing to not only tumor invasion and metastasis just also pharmacokinetic and pharmacodynamics resistance [65].
In studies targeting solid tumors, Machine-T cells surmount challenges conferred by anti-inflammatory factors (e.g., TGF-β and IL-10), immune suppressor cells (Tregs, Bregs, myeloid cells, neutrophils, macrophages), antigen loss, tissue-specific alterations, etc., [51, 64]. Even if Automobile-T cells successfully trafficked to the tumor sites, tumor microenvironment might suppress or even inactivate them. Therefore, with the aim to equip CAR-T cells with capabilities to remodel the suppressive tumor microenvironment by secreting anti-cancer cytokines, strategies named TRUCK have been extensively utilized in CAR-T studies (Fig. 3c). To date, several cytokines take been adopted into TRUCK designs, including IL-12 [66, 67], IL-xviii [68], and TNFRSF14 [69]. Recently, a leading-border TRUCK system using synNotch receptor was described by Roybal et al. [70, 71], which was engineered to produce a range of specific payloads in response to the target antigens. In addition to inflammatory cytokines, the flexible synNotch system could also express pro-tumor cytokine antibodies, checkpoint antibodies, bispecific antibodies, and adjuvants. Using synNotch TRUCK system, more antitumor cytokines or other factors with the potential to reform the local environment based on the specific tumor heterogeneity might exist exploited. For instance, reported as potential anti-cancer therapeutic candidates [72], cytokines such every bit IL-24 should exist further tested.
Anti-immunosuppression
Immunosuppression is a great challenge to effective Automobile-T therapy, equally it enables the tumor cell to escape from antitumor immune responses [73]. Various types of strategy accept been exploited to engineer Motorcar-T cells to fight tumor immunosuppression. Manipulation of the endogenous TCR/MHC (Fig. 3d) and consecration of additional allowed checkpoint receptors (Fig. 3e) are two promising strategies investigated extensively.
Allowed escape mediated by inhibitory pathways via the interaction of activated killer T cell receptors with their ligands, such as programmed cell death1 (PD-1) [74], and cytotoxic T lymphocyte antigen iv (CTLA-4) on T cells is some other major factor [75]. To overcome repressive solid tumor environments and raise the activity and persistence of CAR-T cells, combined therapies using co-administration of immune checkpoint inhibitors or cytokines with CAR-T cells take been employed. Immune checkpoint inhibitors block the immune checkpoint pathways by targeting key regulators in the pathways (e.g., PD-1/PD-L1, CTLA4) to raise the allowed activity of patients' effector T cells [76]. The addition of anti-PD1 monoclonal antibody has been shown to mute the inhibitory effect of such receptors and enhance the function of CAR-T cells in preclinical models [77,78,79]. Many groups are now attempting to generate Machine-T cells resistant to PD1-PDL1 and CTLA4-CD80/CD86 signaling [lxxx], and some of these Machine constructs are currently nether investigation in clinical trials equally discussed in the previous section (Tabular array 1).
Immunosuppressive soluble factors, similar TGF-β and IL10, accept been demonstrated to inhibit CAR-T jail cell activities [81]. Specifically, TGF-β has straight negative effects on T cell differentiation and cytotoxic function, thus hampering T cell effector functions [82]. TGF-β and IL10 tin can likewise inhibit antigen-presenting cells, leading to hampered activation of tumor-reactive T cells [83]. Other factors including prostaglandin E2 (PGE2) and adenosine accept likewise been demonstrated to inhibit T jail cell proliferation and differentiation via signaling through Yard-coupled receptors [84, 85]. Motorcar-T cells can be potentially engineered to include the expression of a ascendant-negative grade of the receptor of these factors to overcome their inhibitory furnishings [86].
In addition to the soluble immunosuppressive factors, various suppressive surveilling allowed cells within the tumor microenvironment, such as Tregs, myeloid-derived suppressor prison cell (MDSC), and tumor-associated macrophage (TAM)/tumor-associated neutrophils (TAN) with the and then-chosen M2 and N2 phenotype, present another obstruction confronting successful CAR-T handling. Tregs have been shown to inhibit T cell activity through cell-to-jail cell contact inhibition and via soluble factors such every bit TGF-β and IL10 [87]. Tregs hamper T cell activity by producing TGF-β, IL10, and also other suppressive agents like IL35 and adenosine [88]. MDSC, M2-TAM, and N2-TAN inhibit antitumor immune response by producing TGF-β, PGE2, reactive oxygen/nitrogen species, and arginase [89, 90]. M2-TAM can express loftier levels of PD-L1, which tin can interact with PD1 on CAR-T cells and inhibit them [91]. To overcome the event of such suppressive surveilling immune cells, the CAR-T design has been proposed to target both the tumor cells and immune cells. Ruella et al. demonstrated the feasibility of targeting CD123-positive Hodgkin lymphoma cells and TAM [92]. However, whether this strategy is universally applicable to other malignancies, especially for the solid tumor, even so needs to be further tested.
Elevating recognition specificity
The genetic heterogeneity of solid tumors is a major outcome to care for such malignancies with Automobile-T. Unlike CD19 in B cell leukemia, at that place is no such "panacea" antigen available for solid tumors. Worse even so, cross-reactions (i.east., "OFF target" and "ON target OFF tumor") with eyewitness non-tumor cells take been widely observed in CAR-T studies, leading to severe or even lethal adverse furnishings caused past T cells attack to the essential tissues.
This off-tumor toxicity could be restrained by designing CAR-T cells with enhanced specificity using two or more than extracellular antigen recognition motifs. Presently, three major classes of bispecific CARs take been employed in T cell technology, namely, dual CAR (Fig. 3f), tandem Auto (TanCAR) (Fig. 3g), and inhibitory CAR (iCAR) (Fig. 3h). Kloss et al. firstly reported co-expression of a suboptimal CAR with an additional chimeric costimulatory receptor (CCR), which sufficiently recognized and killed cells expressing the two antigens [93]. Dual T cells would exist fully activated only when the ii target antigens present simultaneously on tumor prison cell surface, which significantly strengthened the specificity and thus leaving the bystander jail cell untouched. Such dual CAR design was reported to generate specific toxicity towards tumor cells [94]. Recently, an advanced AND-gate circuit using synNotch receptors further upgraded the dual CAR organization [70, 95]. Upon activation by associating with the first antigen, synNotch system induced a secondary CAR expression via intracellular transcriptional domain to modulate T cell action in the presence of the second antigen. Interestingly, initially designed as an AND-gate circuit, a TanCAR that linked 2 distinct scFVs was adopted in treatments against HER2-positive glioblastoma [96, 97], nevertheless has been reported to role as an OR-gate circuit [98], which could kill either CD19-positive or CD20-positive leukemic cells in vivo. This TanCAR has been proved to be particularly useful in the clinic to forestall resistance caused by loss of antigens. To further minimize the T cell "on-target off-tumor" activity towards normal tissues, a Not-gate excursion using a killing Motorcar and an inhibitory CAR was adult [99].
Improving safety and control
About of the efforts at CAR-T engineering has been devoted to improve the strength of clinical response and to prolong T jail cell proliferation and persistence. Yet, the agin effects observed in human being trials suggest the urgency of additional consideration on safety control mechanisms. Utilization of control systems should be regarded as the priority in the next generation Auto-T therapy design every bit T cells are autonomous without regulatory mechanisms.
To engagement, a growing number of user-control systems take been adult to attune CAR-T jail cell expression. In 1997, Bonini et al. firstly reported successful command of graft-versus-host affliction (GVHD) using a suicide factor named herpes simplex virus thymidine kinase (HSV-tk) in adoptive T cell therapy, which rendered the T cells susceptible to ganciclovir handling [100]. In 2001, Fas intracellular domain (ΔFas) was practical every bit a T cell suicide switch to gainsay GVHD in marrow transplantation [101]. In 2011, Di Stasi and colleagues introduced a human caspase-ix (iCasp9) as an off-switch to conditionally trigger apoptosis of CAR-T cells by dimerization upon the handling of small molecules (Fig. 3i) [102]. In hematopoietic stem cell transplantation recipients, xc% of iCasp9-modified T cells could exist eliminated within xxx min nether AP1903 administration without astringent agin furnishings or recurrence [103]. Synthetic death control switches are likely to be extensively utilized in future adoptive immunotherapy owing to their loftier effectiveness, easy controllability, short time of on-set, and mild adverse effects, despite that minority of the T cell populations could escape from the apoptotic signal, resulting in persistent cytotoxicity [103]. Alternatively, CAR-T cells could be removed via apoptosis using antigen-specific monoclonal antibodies (Fig. 3j), such as rituximab for CD20 epitope [104], and cetuximab for EGFR epitope [105]. In add-on to selective emptying, expression of previously known antigens enables tracking of CAR-T cells in vivo.
In contrast to the death switch, a distinct class of on-control mechanisms is considered every bit safer manipulating strategies, as Car-T cells using on-switches are defaulted to exist unresponsive. In this design, CARs are conditionally expressed or form a functional construction in the presence of the inducer, removal of which terminates the transcription or inactivates CARs. Wu et al. described an advanced on-switch Machine using a split construct in vivo (Fig. 3k). The dissever CAR design distributed into two divide polypeptides, an extracellular binding domain scFv and a downstream signaling chemical element ITAM, ii parts reassembled and activated nether the treatment of a modest molecule rapalog [106]. In 2016, Morsut and colleagues farther adult a tissue ligand-specific on-switch CAR construct based on synNotch receptors using a tetracycline (Tet)-regulated promoter (Fig. 3l) [107]. It is noteworthy that expression levels of CARs are generally dose-dependent before saturation, which allows manipulating the strength of Motorcar expression accordingly. In Tet-on systems, CARs still exist after removal of the inducer; hence, information technology requires farther depletion to eliminate Motorcar-T cells ultimately. At this phase, an antibody similar myc could exist conveniently applied to target the tagged CARs as a safeguard factors after adoptive transfer [108]. In principle, integration of the on- and off-switch systems enables us to turn on the expression of CARs at the wanted time point, to turn off when the doses are sufficient, and to erase T cells from patients in one case the treatments are completed.
Strategies involving codon optimization [109,110,111] or construct alteration using lentivirus [112], transposon [113], or RNA [114] were besides employed in the next generation CAR design to improve side effects resulted from T cell overactivation. Hopefully, a combination of suicide/on switches, optimized coding, relevant downstream feedback response, and sensor circuits in the hereafter trials could help to found global control systems to modulate the expression, force, and timing of the engineered Car-T cells.
Determination
Auto-T has demonstrated itself equally a promising handling for the solid tumor in preclinical and clinical studies. Progress in the following aspects of Car-T should facilitate the development of the therapy. Outset, CAR designs need to be further optimized to give improve T jail cell activation, recognition specificity, antitumor activeness, and safety command. The search for optimal signaling and costimulatory domains volition continue to improve the efficacy of CAR-T therapy. Application of bispecific CAR is a promising way to raise the tumor jail cell recognition specificity, limiting unexpected attack to the normal cells. Considering of the genetic heterogeneity, personalized modification during CAR construction might be needed to deliver the maximum antitumor effect. 2d, identification of the most suitable T cell subset for genetic engineering and the institution of a standard ex vivo T cell processing procedure are critical for producing long-lasting CAR-T cell persistence and memory for optimized antitumor response. Additional modifications to Motorcar-modified T cells might be necessary to overcome immunosuppressive tumor microenvironments. Strategies like the introduction of anti-cancer cytokines (IL12), manipulation of the immune checkpoint signaling (PD1/CTLA4), immunosuppressive soluble factors (TGF-β, IL10), and suppressive surveilling immune cells are aspects worth to be explored. Third, the establishment of standard clinical protocols is needed. Improvements in the CAR-T cell itself volition crave parallel developments in clinical protocol blueprint, including patient preconditioning, cytokine support, and other potential co-administered treatments. Preconditioning of the patient earlier adoptive cell therapy is idea to accept a significant outcome on the immune response, thereby producing a potential therapeutic window for CAR-T jail cell activity. Improvements in CAR blueprint and ameliorate understating of the interaction between tumor and immune organization will help to overcome the hurdles currently limiting the application Auto-T in solid tumor handling.
Abbreviations
- ALL:
-
Acute lymphoblastic leukemia
- BTC:
-
Biliary tract cancer
- CAIX:
-
Carboxyanhydrase-IX
- CARs:
-
Chimeric antigen receptors
- CAR-T cells:
-
Chimeric antigen receptor T cells
- CCA:
-
Cholangiocarcinoma
- CCR:
-
Chimeric costimulatory receptor
- CEA:
-
Carcinoembryonic antigen
- cMET:
-
Hepatocyte growth cistron receptor
- CTLA-4:
-
Cytotoxic T-lymphocyte antigen 4
- EGFR:
-
Epidermal growth gene receptor
- EpCAM:
-
Epithelial jail cell adhesion molecule
- EphA2:
-
EPH receptor A2
- FAP:
-
Fibroblast activation protein α
- FDA:
-
Food and Drug Administration
- FR:
-
Folate receptor
- GD2:
-
Disialoganglioside
- GPC3:
-
Glypican-three
- GVHD:
-
Graft-versus-host disease
- HER2:
-
Human being epidermal growth factor receptor-2
- HSV-tk:
-
Canker simplex virus thymidine kinase
- iCAR:
-
Inhibitory Automobile
- IL:
-
Interleukin
- L1-CAM:
-
L1 cell adhesion molecule
- MDSC:
-
Myeloid-derived suppressor cell
- MPM:
-
Malignant pleural mesothelioma
- MSLN:
-
Mesothelin
- MUC1:
-
Mucin
- NK cells:
-
Natural killer cells
- NSCLC:
-
Non-pocket-size-cell lung carcinoma
- PD-1:
-
Programmed prison cell death-1
- PDAC:
-
Pancreatic ductal adenocarcinoma
- PGE2:
-
Prostaglandin E2
- PR:
-
Partial response
- PSMA:
-
Prostate-specific membrane antigen
- ROR1:
-
Receptor tyrosine kinase-similar orphan receptor 1
- scFV:
-
Unmarried chain variable fragments
- TAA:
-
Tumor-associated antigens
- TAM:
-
Tumor-associated macrophage
- TAN:
-
Tumor-associated neutrophils
- TCR:
-
T prison cell receptor
- Tet:
-
Tetracycline
- Treg cells:
-
Regulatory T cells
- TRUCK:
-
T cell redirected for universal cytokine-mediated killing
- VEGFR:
-
Vascular endothelial growth cistron receptor
References
-
Console OKs Machine T therapy for leukemia. Cancer Discov. 2017;7(9):924.
-
Grupp SA, et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. Due north Engl J Med. 2013;368(16):1509–18.
-
Gross Thou, Waks T, Eshhar Z. Expression of immunoglobulin-T-prison cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci U S A. 1989;86(24):10024–8.
-
Ahmad ZA, et al. scFv antibody: principles and clinical application. Clin Dev Immunol. 2012;2012:980250.
-
Dotti G, Savoldo B, Brenner M. Fifteen years of factor therapy based on chimeric antigen receptors: "are we nearly there still?". Hum Factor Ther. 2009;20(11):1229–39.
-
Hombach A, et al. Human CD4+ T cells lyse target cells via granzyme/perforin upon circumvention of MHC class II restriction past an antibiotic-like immunoreceptor. J Immunol. 2006;177(8):5668–75.
-
Fesnak Advertising, June CH, Levine BL. Engineered T cells: the hope and challenges of cancer immunotherapy. Nat Rev Cancer. 2016;16(9):566–81.
-
Irving BA, Weiss A. The cytoplasmic domain of the T prison cell receptor zeta chain is sufficient to couple to receptor-associated signal transduction pathways. Cell. 1991;64(5):891–901.
-
Haynes NM, et al. Redirecting mouse CTL against colon carcinoma: superior signaling efficacy of single-chain variable domain chimeras containing TCR-zeta vs Fc epsilon RI-gamma. J Immunol. 2001;166(1):182–7.
-
Brocker T, Karjalainen K. Signals through T cell receptor-zeta concatenation lonely are insufficient to prime number resting T lymphocytes. J Exp Med. 1995;181(five):1653–9.
-
Finney HM, et al. Chimeric receptors providing both primary and costimulatory signaling in T cells from a unmarried gene product. J Immunol. 1998;161(6):2791–vii.
-
Shen CJ, et al. Chimeric antigen receptor containing ICOS signaling domain mediates specific and efficient antitumor effect of T cells against EGFRvIII expressing glioma. J Hematol Oncol. 2013;6:33.
-
Maher J, et al. Human being T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRzeta /CD28 receptor. Nat Biotechnol. 2002;twenty(1):lxx–5.
-
Savoldo B, et al. CD28 costimulation improves expansion and persistence of chimeric antigen receptor-modified T cells in lymphoma patients. J Clin Invest. 2011;121(5):1822–half dozen.
-
Kofler DM, et al. CD28 costimulation impairs the efficacy of a redirected t-prison cell antitumor attack in the presence of regulatory t cells which can exist overcome past preventing Lck activation. Mol Ther. 2011;19(four):760–7.
-
Koehler H, et al. CD28 costimulation overcomes transforming growth factor-beta-mediated repression of proliferation of redirected human CD4+ and CD8+ T cells in an antitumor cell attack. Cancer Res. 2007;67(5):2265–73.
-
Pule MA, et al. A chimeric T cell antigen receptor that augments cytokine release and supports clonal expansion of primary human T cells. Mol Ther. 2005;12(5):933–41.
-
Hombach AA, et al. OX40 costimulation by a chimeric antigen receptor abrogates CD28 and IL-two induced IL-10 secretion by redirected CD4(+) T cells. Oncoimmunology. 2012;1(4):458–66.
-
Chmielewski Grand, Hombach AA, Abken H. Of CARs and TRUCKs: chimeric antigen receptor (CAR) T cells engineered with an inducible cytokine to modulate the tumor stroma. Immunol Rev. 2014;257(1):83–90.
-
Chmielewski M, Abken H. TRUCKs: the 4th generation of CARs. Expert Opin Biol Ther. 2015;xv(eight):1145–54.
-
Lopez-Albaitero A, et al. Overcoming resistance to HER2-targeted therapy with a novel HER2/CD3 bispecific antibody. Oncoimmunology. 2017;half-dozen(three):e1267891.
-
Yu S, et al. Chimeric antigen receptor T cells: a novel therapy for solid tumors. J Hematol Oncol. 2017;ten(ane):78.
-
Liu B, Song Y, Liu D. Clinical trials of Machine-T cells in People's republic of china. J Hematol Oncol. 2017;ten(ane):166.
-
Hartmann J, et al. Clinical evolution of CAR T cells—challenges and opportunities in translating innovative handling concepts. EMBO Mol Med. 2017;9(nine):1183–97.
-
Gargett T, Brown MP. The inducible caspase-9 suicide gene system as a "safety switch" to limit on-target, off-tumor toxicities of chimeric antigen receptor T cells. Front Pharmacol. 2014;5:235.
-
Diaconu I, et al. Inducible caspase-9 selectively modulates the toxicities of CD19-specific chimeric antigen receptor-modified T cells. Mol Ther. 2017;25(3):580–92.
-
Chen Due north, et al. Car T-jail cell intrinsic PD-1 checkpoint blockade: a 2-in-ane arroyo for solid tumor immunotherapy. Oncoimmunology. 2017;vi(2):e1273302.
-
Ahmed N, et al. Human epidermal growth gene receptor 2 (HER2)-specific chimeric antigen receptor-modified T cells for the immunotherapy of HER2-positive sarcoma. J Clin Oncol. 2015;33(xv):1688–96.
-
You F, et al. Stage i clinical trial demonstrated that MUC1 positive metastatic seminal vesicle cancer can be finer eradicated by modified anti-MUC1 chimeric antigen receptor transduced T cells. Sci China Life Sci. 2016;59(4):386–97.
-
Brown CE, et al. Bioactivity and safety of IL13Ralpha2-redirected chimeric antigen receptor CD8+ T cells in patients with recurrent glioblastoma. Clin Cancer Res. 2015;21(18):4062–72.
-
Katz SC, et al. Phase I hepatic immunotherapy for metastases report of intra-arterial chimeric antigen receptor-modified T-cell therapy for CEA+ liver metastases. Clin Cancer Res. 2015;21(fourteen):3149–59.
-
Petrausch U, et al. Re-directed T cells for the handling of fibroblast activation protein (FAP)-positive malignant pleural mesothelioma (FAPME-1). BMC Cancer. 2012;12:615.
-
Brown CE, et al. Regression of glioblastoma after chimeric antigen receptor T-jail cell therapy. N Engl J Med. 2016;375(26):2561–ix.
-
Brentjens RJ, et al. Safety and persistence of adoptively transferred autologous CD19-targeted T cells in patients with relapsed or chemotherapy refractory B-cell leukemias. Blood. 2011;118(18):4817–28.
-
Kershaw MH, et al. A phase I report on adoptive immunotherapy using gene-modified T cells for ovarian cancer. Clin Cancer Res. 2006;12(xx Pt 1):6106–15.
-
Lamers CH, et al. Treatment of metastatic renal jail cell carcinoma with CAIX Machine-engineered T cells: clinical evaluation and management of on-target toxicity. Mol Ther. 2013;21(four):904–12.
-
Lamers CH, et al. Treatment of metastatic renal cell carcinoma (mRCC) with CAIX CAR-engineered T-cells––a completed study overview. Biochem Soc Trans. 2016;44(3):951–9.
-
Park JR, et al. Adoptive transfer of chimeric antigen receptor re-directed cytolytic T lymphocyte clones in patients with neuroblastoma. Mol Ther. 2007;15(four):825–33.
-
Beatty GL, et al. Mesothelin-specific chimeric antigen receptor mRNA-engineered T cells induce anti-tumor activity in solid malignancies. Cancer Immunol Res. 2014;2(2):112–20.
-
Maus MV, et al. T cells expressing chimeric antigen receptors can crusade anaphylaxis in humans. Cancer Immunol Res. 2013;1(1):26–31.
-
Beatty GL, et al. Safety and antitumor activity of chimeric antigen receptor modified T cells in patients with chemotherapy refractory metastatic pancreatic cancer. J Clin Oncol. 2015;33(15_suppl):3007.
-
Feng K, et al. Phase I study of chimeric antigen receptor modified T cells in treating HER2-positive advanced biliary tract cancers and pancreatic cancers. Protein Cell. 2017. https://doi.org/10.1007/s13238-017-0440-4.
-
Ahmed N, et al. Autologous HER2 CMV bispecific CAR T cells are safe and demonstrate clinical benefit for glioblastoma in a stage I trial. J Immunother Cancer. 2015;three(Suppl 2):O11.
-
Feng K, et al. Chimeric antigen receptor-modified T cells for the immunotherapy of patients with EGFR-expressing advanced relapsed/refractory non-small prison cell lung cancer. Sci Communist china Life Sci. 2016;59(five):468–79.
-
Feng KC, et al. Cocktail handling with EGFR-specific and CD133-specific chimeric antigen receptor-modified T cells in a patient with advanced cholangiocarcinoma. J Hematol Oncol. 2017;10(one):iv.
-
Louis CU, et al. Antitumor activity and long-term fate of chimeric antigen receptor-positive T cells in patients with neuroblastoma. Blood. 2011;118(23):6050–vi.
-
Tchou J, et al. Safe and efficacy of intratumoral injections of chimeric antigen receptor (Car) T cells in metastatic breast cancer. Cancer Immunol Res. 2017;5(12):1152–61.
-
Junghans RP, et al. Stage I trial of anti-PSMA designer CAR-T cells in prostate cancer: possible role for interacting interleukin 2-T cell pharmacodynamics every bit a determinant of clinical response. Prostate. 2016;76(xiv):1257–lxx.
-
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74.
-
Stuelten CH, et al. Chest cancer cells induce stromal fibroblasts to express MMP-9 via secretion of TNF-blastoff and TGF-beta. J Cell Sci. 2005;118(Pt 10):2143–53.
-
Palucka AK, Coussens LM. The basis of oncoimmunology. Cell. 2016;164(6):1233–47.
-
Friedl P, Alexander S. Cancer invasion and the microenvironment: plasticity and reciprocity. Prison cell. 2011;147(5):992–1009.
-
Newick K, Moon Due east, Albelda SM. Chimeric antigen receptor T-cell therapy for solid tumors. Mol Ther Oncolytics. 2016;iii:16006.
-
Moon EK, et al. Expression of a functional CCR2 receptor enhances tumor localization and tumor eradication by retargeted human T cells expressing a mesothelin-specific chimeric antibody receptor. Clin Cancer Res. 2011;17(14):4719–30.
-
Craddock JA, et al. Enhanced tumor trafficking of GD2 chimeric antigen receptor T cells by expression of the chemokine receptor CCR2b. J Immunother. 2010;33(8):780–8.
-
Schuberth PC, et al. Treatment of malignant pleural mesothelioma by fibroblast activation protein-specific re-directed T cells. J Transl Med. 2013;11:187.
-
Lo A, et al. Tumor-promoting desmoplasia is disrupted by depleting FAP-expressing stromal cells. Cancer Res. 2015;75(14):2800–10.
-
Chinnasamy D, et al. Gene therapy using genetically modified lymphocytes targeting VEGFR-2 inhibits the growth of vascularized syngenic tumors in mice. J Clin Invest. 2010;120(11):3953–68.
-
Caruana I, et al. Heparanase promotes tumor infiltration and antitumor activity of CAR-redirected T lymphocytes. Nat Med. 2015;21(5):524–9.
-
Garin-Chesa P, Old LJ, Rettig WJ. Cell surface glycoprotein of reactive stromal fibroblasts as a potential antibody target in human epithelial cancers. Proc Natl Acad Sci U South A. 1990;87(eighteen):7235–ix.
-
Beyer I, van Rensburg R, Lieber A. Overcoming physical barriers in cancer therapy. Tissue Barriers. 2013;i(1):e23647.
-
Beyer I, et al. Controlled extracellular matrix degradation in breast cancer tumors improves therapy by trastuzumab. Mol Ther. 2011;xix(3):479–89.
-
Qin L, et al. Incorporation of a swivel domain improves the expansion of chimeric antigen receptor T cells. J Hematol Oncol. 2017;10(1):68.
-
Balkwill FR, Capasso M, Hagemann T. The tumor microenvironment at a glance. J Cell Sci. 2012;125(Pt 23):5591–6.
-
Khawar IA, Kim JH, Kuh HJ. Improving drug commitment to solid tumors: priming the tumor microenvironment. J Command Release. 2015;201:78–89.
-
Kerkar SP, et al. Tumor-specific CD8+ T cells expressing interleukin-12 eradicate established cancers in lymphodepleted hosts. Cancer Res. 2010;70(17):6725–34.
-
Pegram HJ, et al. Tumor-targeted T cells modified to secrete IL-12 eradicate systemic tumors without need for prior conditioning. Blood. 2012;119(eighteen):4133–41.
-
Avanzi MP, et al. IL-18 secreting CAR T cells heighten cell persistence, induce prolonged B cell aplasia and eradicate CD19+ tumor cells without need for prior conditioning. Blood. 2016;128(22):816.
-
Boice 1000, et al. Loss of the HVEM tumor suppressor in lymphoma and restoration by modified CAR-T cells. Cell. 2016;167(two):405–18. e13
-
Roybal KT, et al. Applied science T cells with customized therapeutic response programs using synthetic notch receptors. Cell. 2016;167(2):419–32. e16
-
Roybal KT, Lim WA. Constructed immunology: hacking immune cells to expand their therapeutic capabilities. Annu Rev Immunol. 2017;35:229–53.
-
Dent P, et al. MDA-7/IL-24 as a cancer therapeutic: from bench to bedside. Anti-Cancer Drugs. 2010;21(8):725–31.
-
Beatty GL, Moon EK. Chimeric antigen receptor T cells are vulnerable to immunosuppressive mechanisms present within the tumor microenvironment. Oncoimmunology. 2014;3(eleven):e970027.
-
Chevolet I, et al. Characterization of the in vivo immune network of IDO, tryptophan metabolism, PD-L1, and CTLA-iv in circulating immune cells in melanoma. Oncoimmunology. 2015;4(3):e982382.
-
Ewing MM, et al. T-cell co-stimulation past CD28-CD80/86 and its negative regulator CTLA-4 strongly influence accelerated atherosclerosis development. Int J Cardiol. 2013;168(3):1965–74.
-
Hegde UP, Mukherji B. Current status of chimeric antigen receptor engineered T cell-based and immune checkpoint occludent-based cancer immunotherapies. Cancer Immunol Immunother. 2017;66(9):1113–21.
-
Condomines Chiliad, et al. Tumor-targeted man T cells expressing CD28-based chimeric antigen receptors circumvent CTLA-4 inhibition. PLoS One. 2015;x(vi):e0130518.
-
Prosser ME, et al. Tumor PD-L1 co-stimulates primary human CD8(+) cytotoxic T cells modified to express a PD1:CD28 chimeric receptor. Mol Immunol. 2012;51(iii–iv):263–72.
-
John LB, et al. Anti-PD-1 antibody therapy potently enhances the eradication of established tumors past cistron-modified T cells. Clin Cancer Res. 2013;19(twenty):5636–46.
-
Pegram HJ, Park JH, Brentjens RJ. CD28z CARs and armored CARs. Cancer J. 2014;twenty(2):127–33.
-
Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12(4):252–64.
-
Massague J. TGFbeta in cancer. Prison cell. 2008;134(two):215–30.
-
Enblad G, Karlsson H, Loskog AS. CAR T-prison cell therapy: the office of physical barriers and immunosuppression in lymphoma. Hum Cistron Ther. 2015;26(8):498–505.
-
Newick K, et al. Augmentation of CAR T-jail cell trafficking and antitumor efficacy by blocking protein kinase a localization. Cancer Immunol Res. 2016;4(six):541–51.
-
Su Y, et al. Cooperation of adenosine and prostaglandin E2 (PGE2) in amplification of cAMP-PKA signaling and immunosuppression. Cancer Immunol Immunother. 2008;57(11):1611–23.
-
Foster AE, et al. Antitumor activity of EBV-specific T lymphocytes transduced with a dominant negative TGF-beta receptor. J Immunother. 2008;31(v):500–5.
-
Sakaguchi South, et al. FOXP3+ regulatory T cells in the human allowed system. Nat Rev Immunol. 2010;x(seven):490–500.
-
Roychoudhuri R, Eil RL, Restifo NP. The interplay of effector and regulatory T cells in cancer. Curr Opin Immunol. 2015;33:101–11.
-
Gabrilovich DI, Nagaraj S. Myeloid-derived suppressor cells as regulators of the allowed system. Nat Rev Immunol. 2009;nine(3):162–74.
-
Mussai F, et al. Neuroblastoma arginase action creates an immunosuppressive microenvironment that impairs autologous and engineered immunity. Cancer Res. 2015;75(xv):3043–53.
-
Ostuni R, et al. Macrophages and cancer: from mechanisms to therapeutic implications. Trends Immunol. 2015;36(4):229–39.
-
Ruella Thousand, et al. Overcoming the immunosuppressive tumor microenvironment of Hodgkin lymphoma using chimeric antigen receptor T cells. Cancer Discov. 2017;7(ten):1154–67.
-
Kloss CC, et al. Combinatorial antigen recognition with balanced signaling promotes selective tumor eradication past engineered T cells. Nat Biotechnol. 2013;31(one):71–five.
-
Wilkie S, et al. Dual targeting of ErbB2 and MUC1 in breast cancer using chimeric antigen receptors engineered to provide complementary signaling. J Clin Immunol. 2012;32(v):1059–70.
-
Roybal KT, et al. Precision tumor recognition past T cells with combinatorial antigen-sensing circuits. Prison cell. 2016;164(4):770–ix.
-
Grada Z, et al. TanCAR: a novel bispecific chimeric antigen receptor for cancer immunotherapy. Mol Ther Nucleic Acids. 2013;2:e105.
-
Hegde K, et al. A bispecific chimeric antigen receptor molecule enhances T cell activation through dual immunological synapse formation and offsets antigen escape in glioblastoma. J Immunother Cancer. 2015;3(Suppl two):O3.
-
Zah E, et al. T cells expressing CD19/CD20 bispecific chimeric antigen receptors preclude antigen escape by malignant B cells. Cancer Immunol Res. 2016;4(6):498–508.
-
Fedorov VD, Themeli M, Sadelain Chiliad. PD-1- and CTLA-four-based inhibitory chimeric antigen receptors (iCARs) divert off-target immunotherapy responses. Sci Transl Med. 2013;5(215):215ra172.
-
Bonini C, et al. HSV-TK cistron transfer into donor lymphocytes for control of allogeneic graft-versus-leukemia. Science. 1997;276(5319):1719–24.
-
Thomis DC, et al. A Fas-based suicide switch in human T cells for the treatment of graft-versus-host disease. Claret. 2001;97(five):1249–57.
-
Di Stasi A, et al. Inducible apoptosis every bit a safety switch for adoptive cell therapy. N Engl J Med. 2011;365(eighteen):1673–83.
-
Zhou 10, et al. Long-term consequence after haploidentical stalk cell transplant and infusion of T cells expressing the inducible caspase 9 safety transgene. Blood. 2014;123(25):3895–905.
-
Vogler I, et al. An improved bicistronic CD20/tCD34 vector for efficient purification and in vivo depletion of cistron-modified T cells for adoptive immunotherapy. Mol Ther. 2010;18(7):1330–8.
-
Wang 10, et al. A transgene-encoded cell surface polypeptide for selection, in vivo tracking, and ablation of engineered cells. Blood. 2011;118(v):1255–63.
-
Wu CY, et al. Remote command of therapeutic T cells through a small molecule-gated chimeric receptor. Science. 2015;350(6258):aab4077.
-
Morsut 50, et al. Engineering science customized cell sensing and response behaviors using synthetic notch receptors. Cell. 2016;164(4):780–91.
-
Kieback East, et al. A safeguard eliminates T jail cell receptor gene-modified autoreactive T cells after adoptive transfer. Proc Natl Acad Sci U S A. 2008;105(2):623–8.
-
Kuball J, et al. Facilitating matched pairing and expression of TCR bondage introduced into human T cells. Blood. 2007;109(6):2331–8.
-
Cohen CJ, et al. Enhanced antitumor activeness of T cells engineered to limited T-cell receptors with a 2d disulfide bond. Cancer Res. 2007;67(viii):3898–903.
-
Provasi Eastward, et al. Editing T cell specificity towards leukemia by zinc finger nucleases and lentiviral gene transfer. Nat Med. 2012;18(5):807–15.
-
Porter DL, et al. Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. Northward Engl J Med. 2011;365(8):725–33.
-
Jin Z, et al. The hyperactive sleeping dazzler transposase SB100X improves the genetic modification of T cells to express a chimeric antigen receptor. Gene Ther. 2011;eighteen(9):849–56.
-
Singh North, Barrett DM, Grupp SA. Roadblocks to success for RNA CARs in solid tumors. Oncoimmunology. 2014;iii(12):e962974.
-
Lamers CH, et al. Gene-modified T cells for adoptive immunotherapy of renal jail cell cancer maintain transgene-specific immune functions in vivo. Cancer Immunol Immunother. 2007;56(12):1875–83.
-
Lamers CH, et al. Allowed responses to transgene and retroviral vector in patients treated with ex vivo-engineered T cells. Blood. 2011;117(1):72–82.
-
Lamers CH, et al. Treatment of metastatic renal cell carcinoma with autologous T-lymphocytes genetically retargeted against carbonic anhydrase IX: get-go clinical experience. J Clin Oncol. 2006;24(13):e20–2.
-
Abate-Daga D, et al. A novel chimeric antigen receptor against prostate stem cell antigen mediates tumor destruction in a humanized mouse model of pancreatic cancer. Hum Gene Ther. 2014;25(12):1003–12.
-
Morgan RA, et al. Instance report of a serious adverse effect following the assistants of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol Ther. 2010;18(4):843–51.
-
Morgan RA, et al. Recognition of glioma stem cells by genetically modified T cells targeting EGFRvIII and development of adoptive cell therapy for glioma. Hum Factor Ther. 2012;23(x):1043–53.
-
Saied A, et al. Neutrophil:lymphocyte ratios and serum cytokine changes later hepatic artery chimeric antigen receptor-modified T-cell infusions for liver metastases. Cancer Gene Ther. 2014;21(xi):457–62.
-
Katz SC, et al. Regional Motorcar-T cell infusions for peritoneal carcinomatosis are superior to systemic delivery. Cancer Gene Ther. 2016;23(5):142–8.
-
Golubovskaya V, et al. CD47-CAR-T cells effectively kill target cancer cells and block pancreatic tumor growth. Cancers (Basel). 2017;9(10):139.
-
Pule MA, et al. Virus-specific T cells engineered to coexpress tumor-specific receptors: persistence and antitumor activity in individuals with neuroblastoma. Nat Med. 2008;xiv(11):1264–seventy.
-
Heczey A, et al. Invariant NKT cells with chimeric antigen receptor provide a novel platform for safe and effective cancer immunotherapy. Blood. 2014;124(18):2824–33.
-
Tanaka Yard, et al. Vaccination targeting native receptors to heighten the office and proliferation of chimeric antigen receptor (Machine)-modified T cells. Clin Cancer Res. 2017;23(fourteen):3499–509.
-
Stroncek DF, et al. Elutriated lymphocytes for manufacturing chimeric antigen receptor T cells. J Transl Med. 2017;15(1):59.
-
Zuccolotto G, et al. PSMA-specific Machine-engineered T cells eradicate disseminated prostate cancer in preclinical models. PLoS One. 2014;9(10):e109427.
-
Gohil SH, et al. An ROR1 bi-specific T-jail cell engager provides effective targeting and cytotoxicity confronting a range of solid tumors. Oncoimmunology. 2017;half-dozen(vii):e1326437.
-
Koneru One thousand, et al. A stage I clinical trial of adoptive T jail cell therapy using IL-12 secreting MUC-xvi(ecto) directed chimeric antigen receptors for recurrent ovarian cancer. J Transl Med. 2015;xiii:102.
Acknowledgements
We repent for non being able to cite all the related work due to folio limit.
Funding
This work was supported by the National Natural Scientific discipline Foundation of China (no. 31500657) and the International Collaboration Projects from the Science and Technology Bureau of Sichuan Province (no. 2017HH0097) to Jian Li. Wenwen Li was supported by the Communist china Scholarship Council.
Availability of data and materials
Data sharing is not applicable to this article every bit no datasets were generated or analyzed during the current study.
Author data
Affiliations
Contributions
JL, WL, GK, and QZ conceived the report and wrote the manuscript. JL, WL, KH, and YZ performed the literature search. All authors read and approved the final manuscript.
Respective author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors have read and canonical the final manuscript for publication.
Competing interests
The authors declare that they take no competing interests.
Publisher'due south Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed nether the terms of the Creative Commons Attribution four.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted utilise, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made bachelor in this article, unless otherwise stated.
Reprints and Permissions
About this article
Cite this article
Li, J., Li, Westward., Huang, Thou. et al. Chimeric antigen receptor T cell (Machine-T) immunotherapy for solid tumors: lessons learned and strategies for moving forward. J Hematol Oncol eleven, 22 (2018). https://doi.org/10.1186/s13045-018-0568-6
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/s13045-018-0568-6
Keywords
- CAR-T
- Solid tumor
- Tumor microenvironment
- Antigen recognition specificity
- Condom control
crenshawbaccustelic.blogspot.com
Source: https://jhoonline.biomedcentral.com/articles/10.1186/s13045-018-0568-6
0 Response to "Editorial Car T Cells - What Have We Learnt? Nature Reviews Clinical Oncology 15 1 (2017)"
Mag-post ng isang Komento